|
Ariane was immersed in antimicrobial resistance research on gram negatives in Ecuador prior to starting an internal medicine residency in 2022 at Salem Hospital and MGB. In her spare time during her medical training, she will be continuing her investigation of gram-negative resistance mechanism in the Kirby lab.
BAARN would not be complete without antimicrobial medicinal chemistry. Roman Manetsch and lab were there in force to highlight collaborative efforts on the streptothricin scaffold.
Some Press Releases:
Researchers Show an Antibiotic Discovered 80 Years Ago Is Effective Against Multi-Drug Resistant Bacteria Neglected 80-year-old antibiotic is effective against multi-drug resistant bacteria Vergeten antibioticum kan uitkomst zijn in strijd tegen multiresistente bacteriën 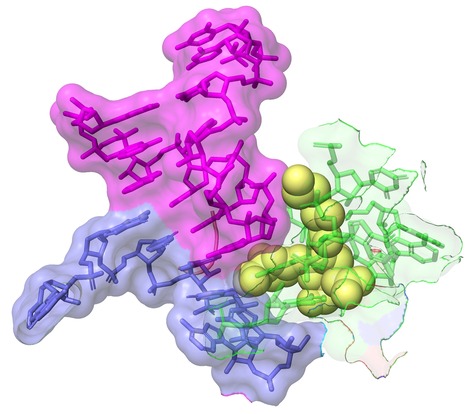
Streptothricin-F (yellow spheres) bound to 16S rRNA (green) of the bacterial ribosome impinges on the decoding site where the anti-codon stem loop of the A-site tRNA (purple) binds to the codon of the mRNA (blue). This bumping of streptothricin-F into the decoding site is associated with translational infidelity (insertion of amino acids into the growing peptide chain other than those encoded for by the mRNA), ultimately leading to poisoning and death of the bacterial cell. The image was created using Pymol by alignment of PDB 7UVX containing streptothricin-F (this manuscript) with the E. coli 70S ribosome from PDB 7K00 containing mRNA and the A-site tRNA shown (DOI: 10.7554/eLife.60482).
General areas include antimicrobial resistance, antimicrobial discovery, novel methods of treating antimicrobial resistant pathogens.
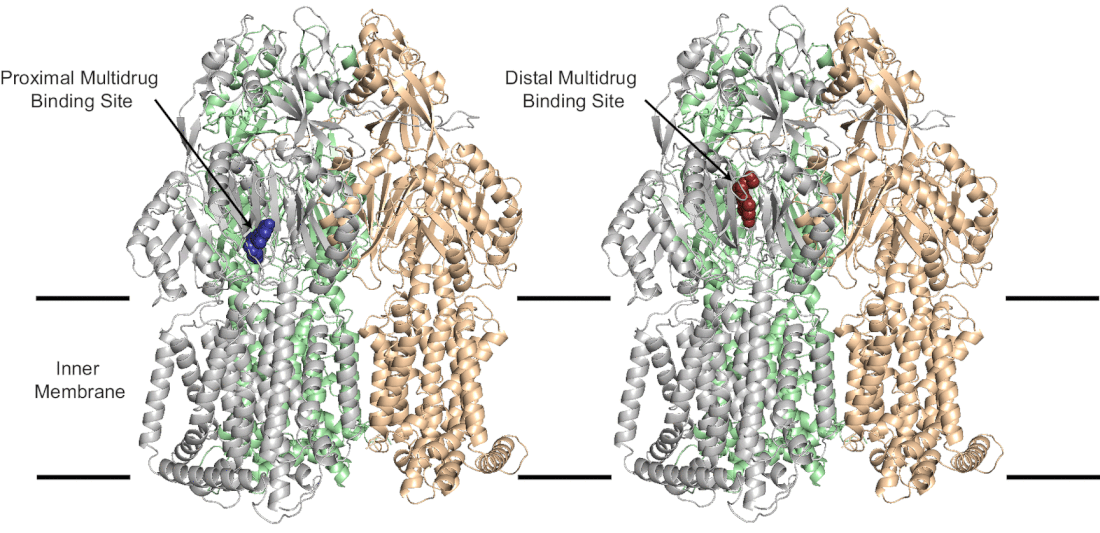
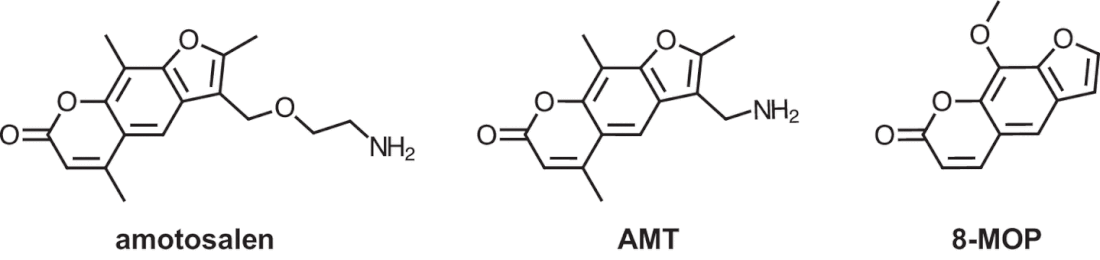
A postdoctoral position is available immediately (4/2/2023) in the Laboratory of Ed Yu to study biology of the ribosome; mechanism of action of translational inhibitors; and interaction of small molecules with RNA in the Department of Pharmacology at Case Western Reserve. Projects are being performed collaboratively with the Kirby, Manetsch, and O'Doherty Laboratories. See recent manuscripts (PMC8805024, PMC8263017, PMC6974574) The incoming research fellow will use x-ray crystallography/cryo-EM and other biophysical/biochemical techniques to elucidate the molecular mechanisms in these systems. Please find additional information about the Yu group and the outstanding training environment in the Case Western Department of Pharmacology website (https://case.edu/medicine/pharmacology/). This position requires expertise in structural biology. The ideal candidate will have shown the ability to guide a project to completion, from protein expression to structural analysis. Knowledge of common structural programs (Phenix, Coot, Pymol) a must. Experience with cryo-electron microscopy sample preparation/operation/data analysis is highly desirable. Experience with single-molecule FRET and other biochemical and biophysical characterization methods would also be considered a strength. Qualified applicants must have a PhD or MD/PhD degree; strong written, oral communication, and critical thinking skills are required. As a Postdoctoral Scholar, he/she will present data at departmental seminars, prepare manuscripts, and contribute preliminary data for grant proposals. Preference will be given to applicants with a recognized record of accomplishment as evidenced by scholarly publications in the field of bacteriology or structural biology. Send inquiries containing a cover letter, CV with list of publications and contact information to Dr. Edward Yu (ewy5@case.edu). See also Ed' posting Just published in mSphere! Images from our manuscript show below. Modeling of amotosalen efflux from the A. baumannii AdeB efflux pump. Ref:
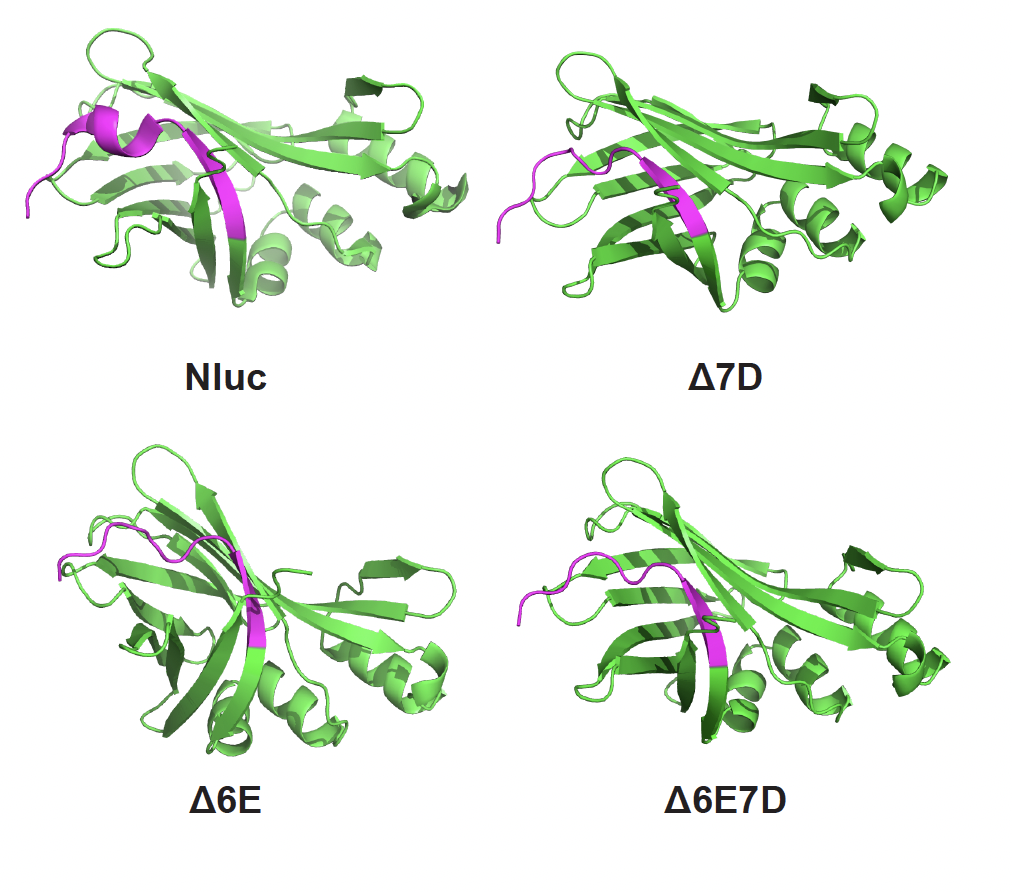
DOI: https://doi.org/10.1128/msphere.00673-22 A Versatile Nanoluciferase Reporter Reveals Structural Properties Associated with a Highly Efficient, N-Terminal Legionella pneumophila Type IV Secretion Translocation Signal just published in Microbiology Spectrum. Figure below is from supplementary data, native alpha-helical N-terminus is a potent T4SS-translocation signal; unstructured N-terminus is not. DOI: https://doi.org/10.1128/spectrum.02338-22
|
Kirby Lab Blog Categories
All
Archives
January 2024
|



 RSS Feed
RSS Feed