|
Our latest manuscript: "Synergistic combinations and repurposed antibiotics active against the pandrug-resistant Klebsiella pneumoniae Nevada strain." Antimicrobial Agents and Chemotherapy early release identifies:
Highly synergistic activity of several antimicrobial combinations. Low apramycin and spectinomycin MIC values. CDC previously reported that none of 26 antimicrobials they tested were active against the Nevada strain. That made the strain until now pandrug-resistant. Links to several news articles about this pandrug-resistant strain:: The Atlantic. NPR, NBC, PBS. Forbes
0 Comments
Adopted by the CDC .in their Antimicrobial Resistance Laboratory Network based on our prior publications in Journal of Clinical Microbiology, Journal of Antimicrobial Chemotherapy, and Antimicrobial Agents and Chemotherapy for testing of aztreonam/ceftazidime-avibactam combination activity using inkjet printing. We provide a step by step video demonstration of assay setup in the Journal of Visualized Experimentation. Link to article landing page (PDF protocol access and video), "Antimicrobial Synergy Testing by the Inkjet Printer-assisted Automated Checkerboard Array and the Manual Time-kill Method" by Thea Brennan-Krohn and James E. Kirby.
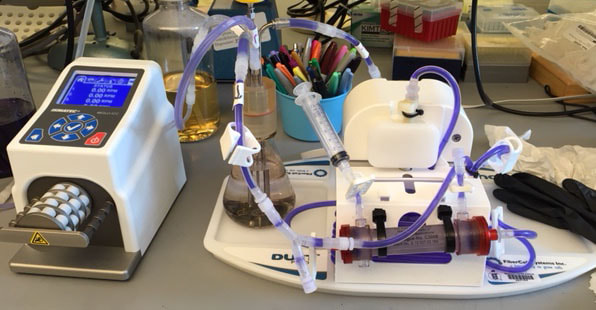
Gentian violet is diluted in hollow fiber cassette with a half life of approximately 4 hours based on dilution of initial "dose" in central compartment with fresh media at flow rate selected on peristaltic pump. Next two antibiotics with different half lives. Will the bacteria outside the hollow fibers survive and for how long?
New antibiotics that may offer additional and much needed treatment options will only be used in hospital systems if the clinical microbiology laboratory can provide timely antimicrobial susceptibility testing data. Historically there has been a time lag in the availability of susceptibility testing methods either at reference laboratories (long delay to results), incorporation in automated commercial systems (4 years), or simpler methods like disk diffusion and gradient strips that can be performed manually on an as needed basis. I was excited to learn that delafloxacin and meropenem/vaborbactam disk diffusion and gradient strip methods finally became available. Fantastic. However, before we can introduce those methods in the clinical laboratory, we need to validate performance of these methods per CLIA '88 regulations and good laboratory practice. This requires either comparing the new methods to a reference standard (broth microdilution -- need antibiotic powder, and a lot of set up time) or a set of strains that has been previously characterized by a reference dilution method and has a good representation of susceptible and resistant isolates. Those are serious roadblocks. By chance, I happened to give a talk at the Northeast Branch of the American Society of Microbiology and a someone from Melinta Pharmaceuticals happened to be there, and that someone referred me to our local Key Account Manager who offerred a solution "on request". Specifically, Melinta or other pharmaceutical companies are not allowed to approach me and tell me about a solution, but if I inquire independently and ask for a previously characterized set of bacterial isolates, they are allowed to tell me that in fact they have a series of previously characterized isolates for both drugs available for validation. These isolates are provided free of charge from Laboratory Specialist, Inc, Sent by FEDEX with first class documentation, data for broth microdilution performed circa ten times on each isolates with modal MICs and MIC distribution - wow, that is awesome. Methods validated beautifully. It just seems odd to me that such a valuable resource, providing clinical laboratories the ability to robustly evaluate AST methods for newly marketed drugs, needs to remain on a need to know basis. Clinical labs, please take note of this available resource. The same proved to be true for plazomicin. Achaogen uses the same Laboratory Specialists, Inc. to provide 30 characterized isolates on request; however, the FEDEX chargers are not absorbed. The FDA-CDC Biobank also now has a set of isolates characterized for plazomicin susceptibility and I see now also delafloxacin., I suspect the exact same set of organisms. The existing rule restricting such company-clinical laboratory communication about these important resources should be relaxed! We need facile access to validation strains sets to bring new testing into our laboratories in a timely fashion, to facilitate the availability of new antibiotics when they are most needed, and to help support profitability of antibiotic development by pharmaceutical companies.
Postdoctoral fellow, Thea Brennan-Krohn, recently had a manuscript accepted in the Journal of Visualized Experimentation, aka JoVE. The title of the manuscript and link to the abstract are "Antimicrobial Synergy Testing by the Inkjet Printer-Assisted Automated Checkerboard Array and the Manual Time-Kill Method." We have been fielding a lot of questions over the past two years about implementation of inkjet printing antimicrobial susceptibility testing technology and thought it would be useful to share a video of the technique as well as classic time-kill analysis to analyze antimicrobial synergy. We are excited to learn that the CDC has decided to implement the technology in the near future in their Antimicrobial Resistance Laboratory Network (ARLN), initially to test, the combination of ceftazidime-avibactam and aztreonam for activity against multidrug-resistant Gram-negatives.
Thanks to American Society of Microbiology science writer, Julie Wolf, for highlighting our inoculum effect manuscript in the ASM mBiosphere Blog, "A Little Change Can Make a Lot of MIC Difference: the Inoculum Effect and Antibiotic Susceptibility Testing"
Now live on the Antimicrobial Agents and Chemotherapy Webpage: The Inoculum Effect in the Era of Multidrug Resistance: Minor Differences in Inoculum Have Dramatic Effect on MIC Determination.
Our inoculum effect manuscript in the journal Antimicrobial Agents and Chemotherapy highlighted in BIDMC news release.
Superbugs: An Arms Race Against Bacteria by William Hall, Anthony McDonnell and Jim O'Neill6/15/2018 |
Kirby Lab Blog Categories
All
Archives
July 2024
|

 RSS Feed
RSS Feed